Oligonucleotides
Synthesis process
During DNA synthesis, each nucleotide is coupled sequentially (from 3’ to 5’) to the growing chain according to the standard ß-cyanoethyl chemical reactions.
Oligos can be modified in several different ways by utilizing the active groups of the nucleotide or creating nucleotide analogues. Modifications can occur either during or after synthesis.
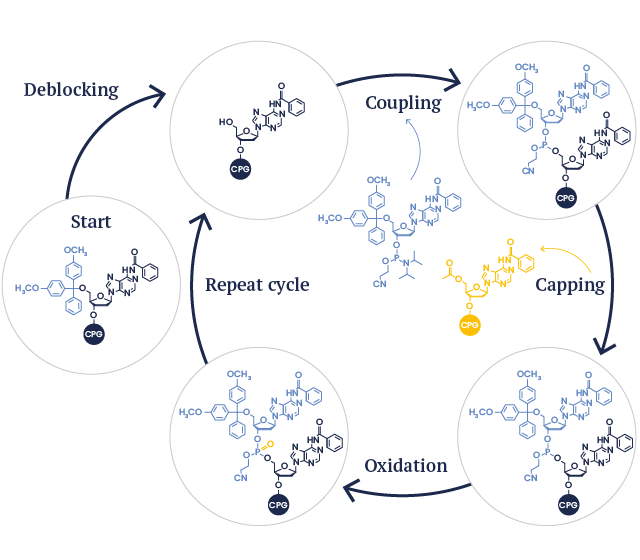
Synthesis Cycle
-
Deblocking
The first nucleotide attached to the solid support is deprotected by removing the DMT-protecting group. This produces a free 5’ hydroxyl group to react with the next nucleotide. -
Coupling
The second nucleotide is activated then added to the reaction and is covalently attached (i.e. coupled) to the previous nucleotide. -
Capping
Any of the first nucleotide that failed to react is capped so that it will no longer participate at any subsequent step. -
Oxidation
The bond between the first nucleotide and the successfully coupled second nucleotide is oxidized to stabilize the growing chain. -
Deblocking
The 5’ DMT group is removed from the second nucleotide to prepare it for further cycles.
At the end of the oligo synthesis, the crude product is cleaved from the solid support (CPG or polystyrene beads) and purified using one of a variety of methods.
Use of amino-modified oligos
In the first case the label is conjugated to an amino-modified oligonucleotide (3’, 5’ or on a dT) using its amino-reactive version (N-hydroxysuccinimide (NHS) ester in most cases).
Use of a maleimide-modified label
The second possibility (originally also used for synthesis of molecular beacons) is the addition of a maleimide-modified label to a thiol-modified oligonucleotide.