Custom Peptides
TR-FRET Peptides
As a world leader in TR-FRET (time resolved Fluorescence) technologies, we are proud to offer you access to custom peptides labeled with lanthanide metals as donor molecules.
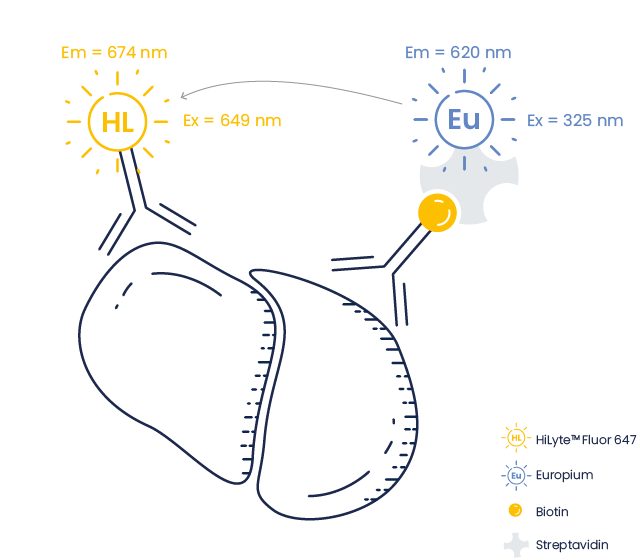
In TR-FRET, instead of a fluorescent dye, the donor molecule is a Lanthanide metal such as Europium (Eu), whose fluorescence is longer-lived than a conventional fluorophore. It is characterized by a large stokes shift, providing a high signal-to-noise ratio due to minimal crosstalk between excitation and emission wavelengths.
TR-FRET allows setting flexible, reliable and sensitive assays, adaptable to high-throughput, with fewer false positive/false negative results.
TR-FRET pairs
The tables below list our TR-FRET dyes and quenchers
| TR-FRET Pairs | |
|---|---|
| Donor | Acceptor |
| Europium (Eu) | HiLyte™ Fluor 647 CyLyte Fluor 5 |
| Europium (Eu) | QXL®610 |
DOTA-Peptide for TR
DOTA is a metal chelator that binds to lanthanide metals.
We can custom synthesize any dota-peptide for use in your other TR applications
Applications
- Interactions between macromolecules (protein-protein; protein-peptide): each interacting partner is coupled to one member of a TR-FRET pair and the energy transfer is measured
- Protease activity measurement: like FRET, TR-FRET can be used to generate peptide which are specific substrates of know proteases
- Protease-based drug target screening and discovery
Why is TR-FRET so sensitive?
TR-FRET is a combination of Time Resolved Fluorescence (TRF) and Forster's (Fluorescent) Resonance Energy Transfer (FRET) technologies.
In TR-FRET the donor molecule is a Lanthanide metal such as Europium. Upon excitation, the energy from the donor (resonance energy) is transferred (donated) to an acceptor (Quencher or fluorescent dye) provided that both molecules are in close proximity.
The acceptor in turn emits light at its corresponding emission wavelength.
Low background with improved assay sensitivity
Biological samples (biological fluids, serum, cells, tissues) typically generate background fluorescence that is detected during a conventional FRET assay, with low signal-to-noise ratio.
With TR (time-resolved) and TR-FRET assays, the fluorescent signal of the lanthanide metals is long-lived and can be measured after the background fluorescence of the biological sample has decayed, resulting in low background with improved assay sensitivity.
Donor fluorophores used in conventional FRET assays have a short stokes shift, with overlapping emission and excitation peaks. This results in background interference from the excitation fluorescence during emission detection. In TR-FRET, the lanthanide donor has a large stokes shift resulting in minimal crosstalk between excitation and emission wavelengths, and a maximal assay sensitivity.
Fig 1. Stokes shift absorption and emission peaks for Europium