Frequently asked questions
Antibodies
Antibodies
How does antibody titer evoluate?
IgMs are more difficult to label and detect than IgGs because of their pentameric structure. To obtain the optimal (highest) IgG/IgM ratio, Eurogentec’s immunization programs are optimized concerning the number and frequency of the boosts.
Special attention was paid to this ratio when developing the Speedy 28-day program, to ensure a minimal amount of IgMs in the final bleed. IgM and IgG antibody titer as a function of time may be represented in the general scheme below.
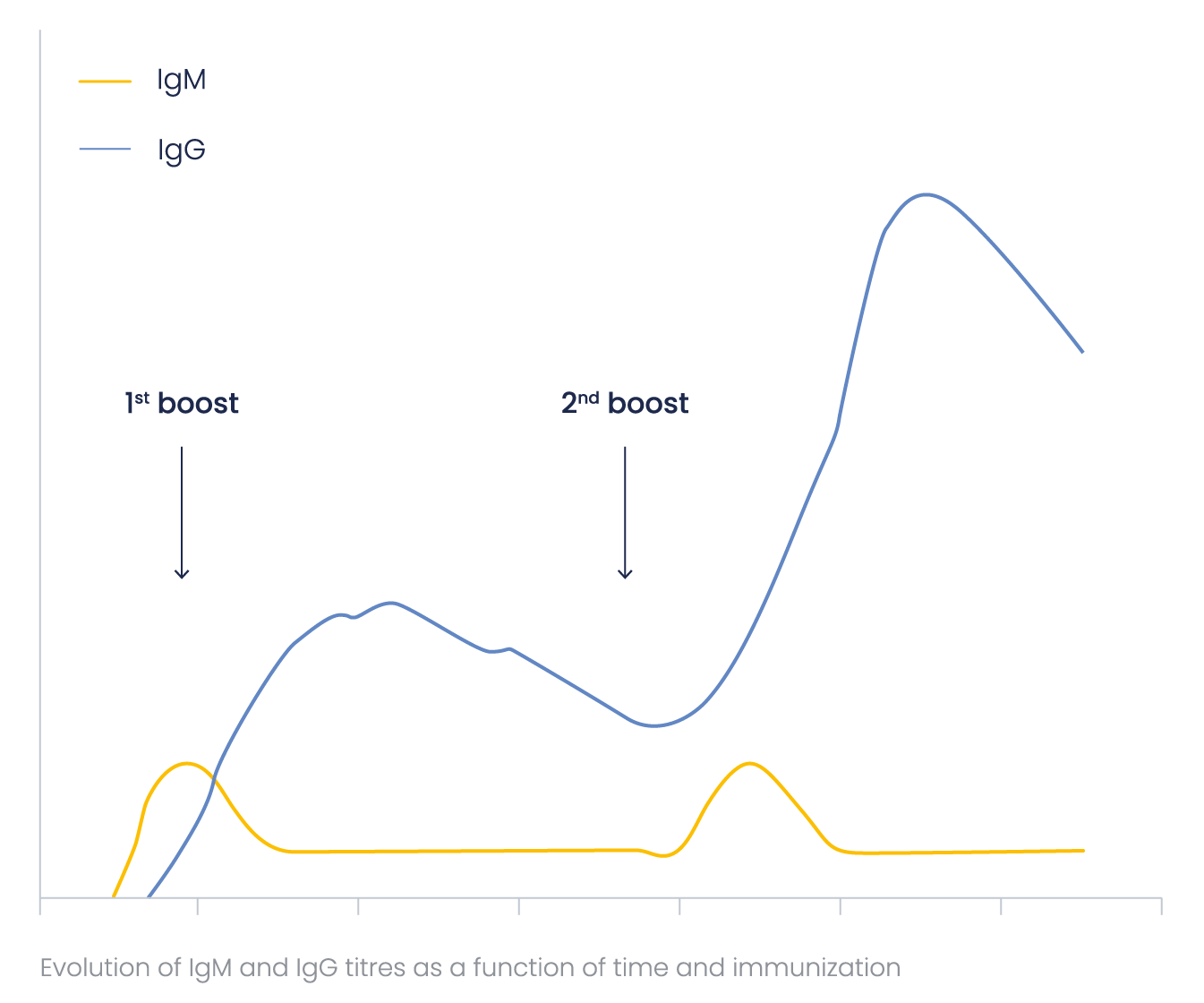
What are the expected bleed volumes?
The immune response depends on the antigen but also on the host. Large animals provide the advantage of allowing larger bleeds but their immune response is generally lower than small animals. The following table indicates the expected bleed volume in various hosts.
| Pre-immune | Small Bleed | Large bleed |
Final bleed | Comment | |
| Mouse | 40-70 μL | 40-70 μL | 40-70 μL | 300-500 μL | Good to test antigenicity |
| Guinea pig |
1 mL | 1 mL | 2-3 mL | 10-15 mL | For small serum volumes |
| Rat | 2 mL | 2 mL | 2 mL | 5 mL | For small serum volumes |
| Chicken | 1 egg | ± 8-10 eggs* | ± 8-10 eggs* | ± 8-10 eggs* | For mammalian antigens and large quantities of Antibodies, 4 eggs = |
| Rabbit | 2 mL | 2 mL | 20-25 mL | 50-70 mL | For most applications |
| Goat | 2 mL | 2 mL | 250 mL | 1000 mL | For large batch volumes |
| Sheep | 2 mL | 2 mL | 250 mL | 1000 mL | For large batch volumes |
| Llama | 2 mL | 2 mL | 250 mL | 1000 mL | Single chain antibodies and for large batch volumes |
How much Antigen do I need for an immunization?
Injection amounts per rabbits depend on the antigen weight:
100 μg per injection for < 18 – 20 kDa proteins*;
200 μg per injection for > 18 – 20 kDa proteins.
Check here the Injection amounts for other hosts.
*Please note that the molecular mass of haptens is usually too low to elicit an immune response. Therefore, they should be provided under a format with a higher MM if Ab production is requested (e.g. coupled to a carrier).
Which animal should I use for antibody production?
The choice of animal is dependent on practical factors such as the source of the antigen, quantity of serum required for research, the titer of the antibody and the time required to generate antibodies. Antigens coming from rabbits should be injected into pigs or chickens rather than into rabbits. Mammalian proteins can be injected into chickens to provide high titer antibodies, which have the advantage of being IgY type antibodies.
Program duration: generally, the smaller the animal the shorter the program duration and the smaller the volumes of serum received. As antigenic response varies from animal to animal of the same species, repeating a immunization program is more risky than choosing a larger animal for antibody production to ensure sufficient material. It is recommended to immunize at least 2 animals to ensure a high quality antibody at the end of the program.
Which animals are available?
Eurogentec offers a very large range of animals, mice, rats, chickens, rabbits, sheep, goats, pigs, llamas, alpacas... Should you require a different host than listed please let us know.
What is an SPF animal?
Specific Pathogen Free animals are bred uniquely for the production of antibodies. These animals are housed in a pathogen free environment – all entering air, food, water is guaranteed to not contain foreign organisms that could provoke an immune response. These animals offer a very low background signal due to the restricted environment in which they live.
How long does antibody production take?
Antibody production can be done in as short a time as 2 months although we recommend 3 months for most hosts.
How many boosts and bleeds do you do?
The immunization program is species dependent, however, in general animals are immunized three times the first month then are given booster shots once a month for as long as is required. Bleeds are done 15 days after immunizations.
What happens if you reuse an animal for a second or third immunization?
The risk of reusing animals for other immunization programs is the presence of antibodies from past immunizations. This can lead to increased background signals and false positive responses in downstream applications.
Is it important to get a terminal / final bleed?
Yes, terminal bleeds ensures that you are not getting serum from a host that has already been used in a previous immunization program, which could potential contaminate your supply of antibodies.
What if my antigen is insoluble in biological buffers?
Protein concentrations should be at least 1mg/ml with minimal amounts of urea, SDS, guanidine hydrochloride (and other chemotropic agents). Should the antigen require the presence of these agents then the antigen concentration should be raised to 5-10 mg/ml. By injecting small volumes (10-50 µl) we are often able to generate an immune response to the antigen.
What buffers are compatible with immunizations?
Most biologically related buffers are compatible with immunization, this includes PBS, Tris buffer, phosphate buffer, in moderate molarity. The antigen should be in a concentration of 1mg/ml or better and should be shipped frozen on dry ice when possible.
How should I ship my antigen?
Samples should be shipped on dry ice whenever possible. Peptides and SDS-PAGE gel slices can be sent at room temperature.
Can I extend my program to get more bleeds and antibodies?
All programs can be extended by adding additional immunizations and monitoring response by ELISA. When sufficient titer is present the final bleed can be ordered.
What is the pre-immune serum for?
Evaluation of pre-immune serum in your application permits the selection of the best animal for the subsequent production of antibodies. The selection criterion is no-signal in the application (eg nothing in Western Blot) due to the presence of previously generated antibodies thus ensuring very low background signal after immunization. Pre-screening is particularly important when making antibodies to against common bacterial, viral or common allergens. We recommend screening 5 or 10 animals and selecting the best for immunization.
How long should my peptide be?
Epitopes are generally 6-8 amino acids in length, by using a peptide of 16 amino acids in length there is the possibility of raising 10 different antibodies against the sequence used. This elevates the success rate of the program, although we have had success using peptides of shorter and longer lengths. Long peptides (>20’mers) are not recommended as these could contain secondary structures not found in the natural antigen.
Epitopes are generally 6-8 amino acids in length, by using a peptide of 16 amino acids in length there is the possibility of raising 10 different antibodies against the sequence used. This elevates the success rate of the program, although we have had success using peptides of shorter and longer lengths. Long peptides (>20’mers) are not recommended as these could contain secondary structures not found in the natural antigen.
How is the peptide conjugated to the carrier protein?
Peptides can be conjugated to a carrier protein via the N-terminus, C-terminus or an internal amino acid (not recommended). Coupling with glutaraldehyde is used to couple the N-terminus of the peptide to the lysine residues (via the amino sidechain) of the carrier protein; coupling with EDCI (1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide) is used to link the C-terminus of the peptide to the lysine residues of the carrier protein; coupling with MBS (m-Maleimidobenzoyl-N-hydroxysuccinimide ester) is used to link a cysteine via its SH group to the lysine residues of the carrier protein. This very common approach involves adding a cysteine residue to the selected peptide sequence prior to coupling.
What is a carrier protein?
A carrier protein is a protein that is used to deliver the peptide in an immunization program. As the peptide’s molecular weight of 1.5 – 2 kDa will not elicit an immune response on its own, the peptide is conjugated to a protein to increase its apparent molecular weight. The host will then generate antibodies against the peptide and the carrier protein.
Which carrier protein should I use?
For best results a carrier protein should elicit a strong immune response. For this reason the carrier protein should be unrelated to the host species.
Eurogentec offers a number of carrier proteins (BSA, OVA, THY), but we find that we get best results from KLH (Keyhole limpet hemocyanin).
What purity of peptide is best for antibody production?
Although high purity peptides are better than impure peptides for the generation of specific antigens, high purity peptides are not required for successful antibody generation. Immunograde peptides have greater than 70 % purity and are routinely used in immunization programs.
Is it possible to make antibodies against non-peptides (haptens)?
In analogy to conjugating peptides, it is possible to conjugate low molecular weight molecules to spacers and then to carrier proteins. In this way antibodies against hormones for example can be generated.
What are MAP peptides?
Multiple Antigen Peptide is an alternative method of generating antibodies from peptides. Rather than conjugating the desired peptide to a carrier protein the peptides are synthesized onto a polylysine resin. The resulting structure, once cleaved from the resin, is a polylysine core surrounded by 8 copies of the peptide with an overall molecular weight of 13-17 kDa, sufficient to elicit an immune response. Literature reports suggest that better immune responses are generated from this approach in some cases.
Purification of antibodies generated by the MAP strategy require the additional synthesis of free peptide for subsequent affinity purification.
What kinds of antibody purifications are available?
There are two main types of purifications; the first is to concentrate the antibodies from the other proteins found in serum, the other is to specifically concentrate the antibody of interest. The first case can be done with protein A or protein G columns. Protein A columns are useful for concentrating polyclonal antibodies from human, rabbit, horse, pig, guinea pig, dog while protein G columns are useful for concentrating polyclonal antibodies from mouse, rat and goat sources. Affinity purification to solely concentrate the antibody of interest can be done by coupling the peptide or the purified protein to a resin. The affinity of the antibody for the antigen will cause the retention of the desire antibody. This is then eluted with a salt or pH gradient.
Is there a way to monitor antibody production during the immunization program?
A common approach for evaluating the presence of the desired antibody is with an ELISA (Enzyme Linked ImmunoSorbent Assay). This involves the coating of a plate with the desired peptide, capturing the relevant antibodies from the serum, washing to retain only the relevant antibodies, detecting with the secondary antibody HRP (horse radish peroxidase) in the presence of a chromophore. This colorimetric assay is used to determine the titer of antibody present in the serum. Eurogentec recommends doing an ELISA after the first bleed.
What happens if the ELISA results are negative?
Negative results are replicated to ensure that the results are reproducible. Confirmation of results is best done by Western blot analysis to confirm the absence of antibodies. In the case of both negative ELISA and Western blot results we would recommend the termination of the program and the selection of a different antigen and/or a different host.
What is an ELISA test?
A common approach for evaluating the presence of the desired antibody is with an ELISA (Enzyme Linked ImmunoSorbent Assay). This involves the coating of a plate with the desired peptide, capturing the relevant antibodies from the serum, washing to retain only the relevant antibodies, detecting with the secondary antibody HRP (horse radish peroxidase) in the presence of a chromophore. This colorimetric assay is used to determine the titer of antibody present in the serum. Eurogentec recommends doing an ELISA after the first bleed.
What is a secondary antibody?
A secondary antibody is an antibody used to detect the presence of the primary antibody. An antigen is used to generate a primary antibody in a host animal, for example in rabbit. To detected and quantify the amount of the primary antibody (ELISA, WB, etc) a secondary antibody is used, in this example an anti-rabbit antibody. Typically, the secondary antibody is conjugated to an enzyme that converts a substrate from a non-colored state to a colored state, providing a colorimetric assay. When used with standard curves one can accurately quantify the amount of primary antibody present in a sample.
How many egg yolks does a standard program yield?
Chickens typically yield an egg a day. A 2 month protocol thus yields approximately 30 eggs, the first month being the month of immunizations.
What is Western blotting?
Is a qualitative technique to detect the presence of an antigen in a sample with an antibody. The process involves extracting all the proteins from a sample, separating the proteins in 1D or 2D polyacrylamide gels, transferring the proteins from the gel to a membrane (PVDF or nitrocellulose), blocking the portions of the gel where there are no proteins (typically with milk), treating the membrane with the antibody, washing away excess antibody, detecting the presence of the antibody with a secondary antibody. This method is dependent on the epitope the antibody recognizes. As gel separation methods are denaturing the best antibodies for Western blotting are those that recognize linear sequences.
What causes background in a Western blot?
Background signals can due to non-specific binding of the primary antibody to proteins other than the antigen; due to the presence of spurious antibodies mixed in with the primary antibody; or poor blocking of membranes.
How should I store my antibody?
Storage Suggestion for Antibodies used for in vitro Research Applications
Properly stored, antibodies should remain stable for months or years. Freezers with automatic de-frosting are absolutely inappropriate for antibody storage.
If you are using catalogue antibodies, please refer to the provided data sheet.
Storage Temperatures
Our purified antibodies are delivered to you in PBS with 0,1% BSA as stabiliser and 0,01% Thimerosal as preservative. For these formulations we suppose that storage of small aliquots at –20°C is appropriate.
The aliquoting of antibodies has the following advantages:
- Minimisation of the damage due to freezing and thawing
- Exclusion of contamination due to multiple pipetting from a single vial
Aliquots should be thawed once, with any remaining antibody kept at 4°C.
When you receive an antibody you should carefully thaw it (in the fridge in a 4°C water bath – do not forget about it) and centrifuge it with 10.000 x g for 20 sec in order to pull down all the solution. Aliquot the antibody in low-protein-binding micro centrifuge tubes for storage. Please adapt the size of the aliquots to your typical experimental consumption. Aliquots should not be smaller than 10 µl to avoid damage of the antibody by evaporation and adhesion to the vessel wall.
Though it is considered in general as optional, we recommend the addition of 50% glycerol (v/v, final) to your antibodies for additional freezing and evaporation protection for storage at –20°C (glycerol is frozen at –80°C).
Please use just sterile chemicals and solutions for your antibodies to prevent down stream microbial growth.
Upon receipt, antibody storage for one or two weeks (time required for experimental establishment) at 4°C should be acceptable with subsequent aliquoted freezing for long-term storage.
Ascites and serum represent an exception here since the contained proteases require immediate storage at -20°C or -80°C.
Please be advised that sera produced at Eurogentec do not contain any preservative, 0,01% azide or thimerosal (depending on your research purpose) should be added to the aliquots for storage at –20°C after first thawing.
Egg yolks in contrast must never be frozen, since they denature into a form that does not allow isolation of IgY and affinity purification anymore. Egg Yolks from Eurogentec are shipped at 4°C with 0,01% azide added as preservative ready for short-term storage (over several weeks) at 4°C.
For long-term storage we recommend the purification of IgY from the egg yolks.
IgY isolates and affinity purified IgY antibodies should be treated like purified antibodies described in the beginning of this section.
Antibody formulations that require special attention:
- Enzyme-conjugated antibodies should not be frozen at all in order to retain the maximum enzymatic activity, storage at 4°C is therefore mandatory, please refer to the technical datasheet on the solvent formulation, and add if required 0,01% Thimerosal (Azide will inactivate HRP-conjugates);
- All conjugated antibodies (enzymes, fluorophores, or biotin) should be stored in the dark (very well suited: the black foil bags of x-ray films or photographic paper, and the corresponding boxing);
- Fluorophore-conjugated antibodies can be stored aliquoted at –20°C with 50% glycerol (final v/v) – please make sure by the data sheet that they are formulated as well with BSA, physiologically concentrated buffer salts (e.g. PBS), and preservatives (azide or thimerosal), furthermore, please be sure to use a sterile glycerol formulation, since microbial growth might occur and destroy the antibody. Storage at –80°C of glycerol containing solutions is not advised since this is below the freezing point of glycerol;
- Minimisation of aggregate formation of some IgG isotypes (IgG3 for instance) requires storage at 4°C.
Contamination prevention
Purified custom-made antibodies from Eurogentec already contain 0,01% thimerosal to avoid microbial growth, as mentioned on the data sheet. But please be advised that any serum, like Test Sera for animal selection, pre-immune sera (PPI), large bleed sera (GP), or final bleed sera (SAB) do not contain any preservative.
Please be advised of the following prior to use sodium azide for storage :
- Azide is toxic to organisms and should be avoided in applications which are done with living cells, or tissues (the same is the case for thimerosal – please advise us before starting your immunization programme of your research purpose in order to avoid any preservative);
- If antibodies should be conjugated or spotted involving amino groups, azide will interfere negatively into the process, thimerosal is an acceptable alternative without these unwanted side effects.
Preservatives, if already in an antibody formulation, can be removed in the following ways:
- By dialysis or centrifugal filtration: using a device with a MWCO of 14 kDa, please refer to the manufacturer’s recommendations for most efficient use centrifugal filter devices or dialysis equipment (MW of sodium azide ~ 65 D, thimerosal ~ 404 D, IgG ~ 160 kDa, IgM ~ 600 kDa);
- By gel filtration (which is faster than dialysis). Please refer to the manufacturer’s recommendations for most efficient use of suited filter cartridges
please be advised that all materials used here should be sterile to ensure the subsequent stability of the antibody, and to avoid contamination by purification.
Damage caused by freezing and thawing of antibodies
The antibody’s binding capacity can be reduced by aggregate formation due to denaturation following cycles of freezing and thawing.
Though a lot of researchers keep their antibodies at –80°C, there is no real explanation why –20°C might be inappropriate. In any case, antibodies should rather be stored protected from daily routine in the freezer, e.g. rather in the back than in the front, or in drawers than standing free accessible.
Working concentrations of antibodies should not be stored for more than one day at 4°C, since proteins are more susceptible to degradation if they are stored in lower concentrations. Ideal would be to formulate the antibody with additional protein, like BSA or milk powder to reach concentrations of total protein in the mg/ml range. BSA or milk powder in working solutions also minimizes antibody loss due to attachment to the vessel wall.
The addition of stabilizing proteins is not recommended for antibodies that are subject to conjugation or array spotting.
What information does the ELISA test provide to you ?
The ELISA test carried out by Eurogentec is an indirect ELISA. This means that a constant amount of antigen has been coated into the wells of the ELISA plate (100 ng/well), and tested with different dilutions of the serum or antibody in question. The development is done colorimetric, using a secondary HRP-conjugated antibody, and o-phenylenediamine as chromogenic substrate. The optical density of the chromogenic substrate is measured at 492 nm OD(492).
The method has to be considered as semi-quantitative, since the measured reactivity with growing dilutions can be due to:
- The concentration of specific antibodies against the antigen
- The affinity of the evolved antibodies against the antigen
The results are influenced by the following factors:
- The general success of the immunization against the antigen
- The suitability of the antigen (peptide or protein) to be coated onto ELISA plates
- The exposure of the immunization relevant epitopes after coating
The measured optical densities at 492 nm are plotted against the dilution of the antibody or serum. The following curve types can be found:
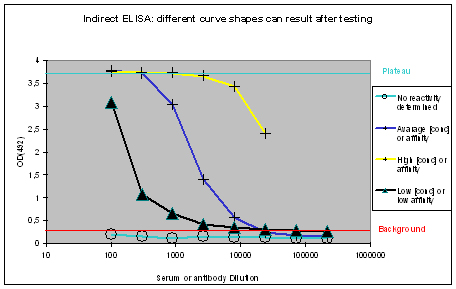
Figure 1 : Different shapes of ELISA curves that can be found after ELISA testing. “No reactivity determined” (light blue), “Low [conc.] or affinity” (black), “Average [conc.] or affinity” (blue), and “High [conc.] or affinity” (yellow) display flat, hyperbolic, or sigmoidal ELISA curve shapes. The optical density OD(492) can be correlated to antibody affinity or concentration in the window between the “Background” (red), and Plateau (turquoise).
Flat ELISA curves
Flat ELISA curves (Figure 1, light blue) are typical for pre-immune sera. Since the immunization host did not encounter the antigen, no immune response could be determined. A flat curve can also result, if antigens coated with the epitope towards the ELISA plate, or which did not coat at all. Potential program difficulties: a flat ELISA curve can result in rare cases, if the antigen is a serum protein, cell surface expressed, and evolutionary conserved – the generation of an immune response will just be suppressed, in order to protect the host (which is normal).
Given all the variables, testing and establishment of the antibody should be carried out accordingly.
Hyperbolic ELISA curves
Hyperbolic ELISA curves (Figure 1, black) are typical for the immune response generation phase during an immunization program. This reactivity kinetics can be found very often when testing the small test bleed (PP) of 87-day programs. The curve might be understood as “a low concentration of high affinity antibodies” has been generated, but it also can result from antigen coating difficulties. Given this, testing the antibody in your application might give completely different results.
Sigmoidal ELISA curves
The first case of sigmoidal ELISA curves represents the average of ELISA tests carried out by Eurogentec. In Figure 1 the curve is represented in blue. The curve falls in a sigmoidal shape from the “Plateau” of saturation usually at a dilution of 1:1.000 towards the “Background”, which is reached with a dilution higher than 1:10.000. The curve might be read like “the immunization provided a mixed population of high and low affine antibodies, which detect the antigen even if highly diluted”. Antibodies or sera producing curves like this can be used in a lot of applications, since the antibody population is rich – the ELISA test only detects antibodies that bind to the epitope immobilised on the ELISA plate, other populations of the antibody are not reflected here.
The second case of sigmoidal ELISA curves is over the average of ELISA tests carried out by Eurogentec. This curve is displayed in yellow in Figure 1. The curve is characterised by keeping the “Plateau” level of saturation very long, and falling in a sigmoidal fashion towards the “Background” level beyond the concentration range considered for ELISA plots. Curves like this result from very immunogenic peptides, large proteins like maybe a provided antigen from customers or the peptide carrier protein KLH used by Eurogentec, or so called “super antigens”. While strong reactivity against your peptide, or protein is desired, and a strong reactivity against KLH is a positive control for general successful immunization using peptides, super antigens represent epitopes that can be found randomly. This random occurrence of structural instead of sequence-related motifs implies as well that the antibody providing this is non-specific. The cases where immunizations provided antibodies to super antigens are rare. Antibodies or sera producing curves like this can usually be used in a lot of applications, since the antibody population is richly composed.
Purifications
After an immunization, you might have included affinity purification of the produced antibodies against the peptide or your antigen in the sufficient amount. Eurogentec offers different formats of purifications, 5 ml, 20 ml and 50 ml. The ELISA can be added optionally to 5 and 20 ml purifications, but it is a part of 50 ml affinity purification. The ELISA has to be considered here, as a mean of characterisation for purified antibodies, and not as a tool providing a quality control. As above already mentioned, the results of the ELISA tests are influenced by
- The accessibility of the epitope for the antibody
- The concentration and plating suitability of the antigen
- Simply the fact that the antibody has been purified with the antigen coupled to a matrix, and is now probed with the antigen immobilised on an ELISA plate (structural differences of the antigen in two different applications)
Given these factors, different results might be possible for the ELISA of the purified antibodies:
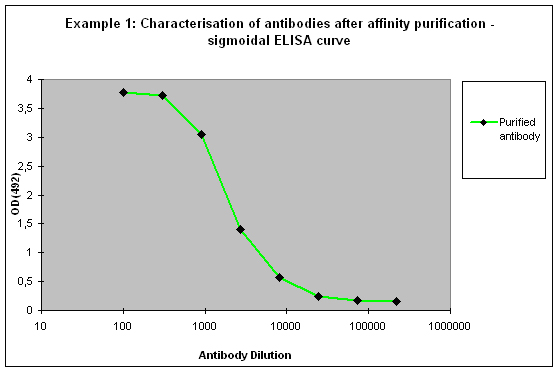
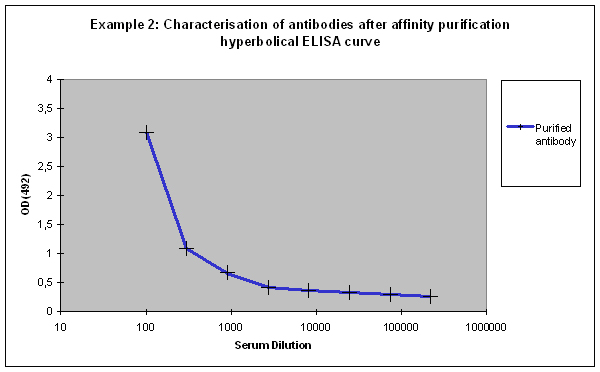
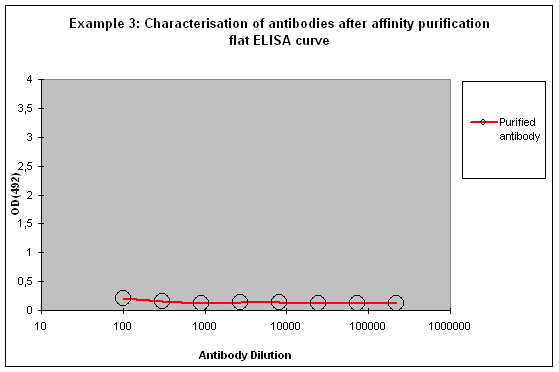
Purification 3 : a flat ELISA curve might suggest that the purification did not provide any working antibody (Example 3). Please be advised that also in such a case testing is worth the effort, since it might still be possible that the antigen looks different on the ELISA plate compared to its structure during affinity purification, and finally in your application. Try to characterise the antibody by Western or dot blotting – these applications work with far over 70% of custom-made antibodies from Eurogentec. A starting dilution might be 1:500. If it tests positive in Western blot – or dot blot, and you want to use your new antibody in your desired application, you have to establish it accordingly, maybe change existing protocols. Please compare the performance of the purified antibody always to the serum (1:500 to 1:10.000 diluted, depending on your optimisation results) in paralleled tests on identical samples. Should you experience difficulties, please feel free to contact us for further advice.
ELISA and Cross Purifications from Phospho-, Methyl-, or other modification specific programs
Since protein modifications play a key role for different cellular functions, and scientific interest for the different types of protein editing events is gaining more attention, Eurogentec is proud to make the detection of post-translational modifications also possible to you.
Immunization programs for their detection can be carried out in rabbits or guinea pig, as Eurogentec’s standard 87-day mode or as 28-day Super Speedy immunizations, but the crucial point is the ELISA testing during, and after the programs.
Identifying the best host for cross affinity purification of modification specific antibodies by ELISA
As in any immunization program carried out by Eurogentec two hosts (in this case rabbits or guinea pigs) have been immunised with a peptide carrying the modified amino acid that you want to detect in down stream experiments.
To identify the best responding host, the pre-immune serum (PPI), and the final bleeds (SAB) of the hosts are tested against the modified peptide and against the carrier protein by indirect ELISA (Figure 2)
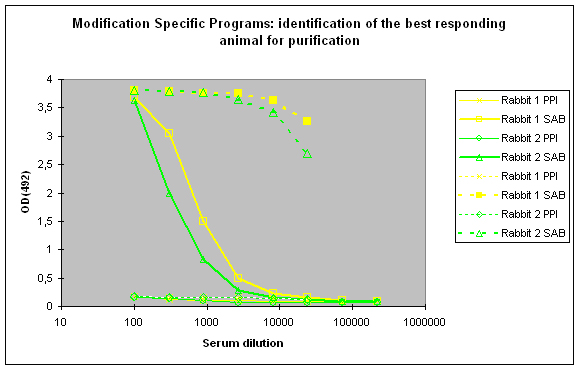
Figure 2 : In this anti-phospho program the immune responses against the phosphorylated peptide (solid lines, rabbit 1, yellow, and rabbit 2, green) and against the carrier protein (dashed lines) are determined by Indirect ELISA (Coated antigen, two hosts, and two bleeds, part of modification specific programs) and compare the reactivity of the serum before immunization (pre-immune serum, PPI, day 0), and the final bleed (SAB, end of the program). The serum to be chosen for cross purification based on Eurogentec’s expertise would be rabbit 1, since the response here is a little bit better compared to rabbit 2 (SAB, solid green and yellow lines). This impression is also supported by the finding that the response from rabbit 1 against the carrier protein (SAB, dashed yellow line) is a little bit better than the carrier response of rabbit 2 (SAB, dashed green line). In analogy to Fig. 1 (above) spoken, both responses to the peptides are good, and similar, but rabbit 1 (SAB, solid yellow line) produced a response a little bit closer to the ideal sigmoidal shaped curve.
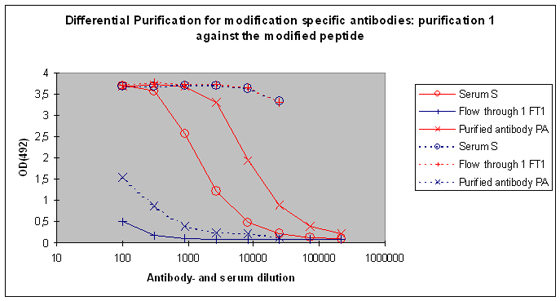
Purification 1 : isolation of antibodies that are specific to the peptide and to the modification from rabbit 1
After the best responding host has been identified by the above-performed indirect ELISA, the final bleed serum (SAB) of this host will be used for cross purification. The first affinity purification will be carried out against the modified peptide. By this step the serum will be depleted from antibodies that are specific to the modification, and the peptide context in general. Following this purification, the serum (S), the flow through 1 (FT1), and the purified (PA) are tested by indirect ELISA against the modified peptide and the carrier protein. In general the following results can be expected (Please see also Figure 5) :
|
Fraction |
Reactivity against mod. peptide |
Reactivity against carrier |
|
Purified antibody (PA) |
Yes, stronger than S, and much stronger than FT1 |
No, if Yes then removal by dilution quickly possible |
|
Flow through 1 (FT1) |
No, if Yes than much weaker than S or PA |
Yes |
|
Serum (S) |
Yes, weaker than PA |
Yes |
Purification 2 : separation of modification specific antibodies from just peptide specific antibodies
Peak 2 and FT2 will be characterised by indirect ELISA against the modified peptide, and against the non-modified peptide. In general the following results can be expected:
|
Fraction |
Reactivity against mod. peptide |
Reactivity against the non-modified peptide |
|
Peak 2 (P2) |
Yes, the modified peptide contains a lot of antigens |
Yes, the modified peptide contains a lot of antigens |
|
Flow through 2 (FT2) |
Yes |
No, if any it can easily be diluted away |
A sample result is reflected in Figure 4 and Figure 5.
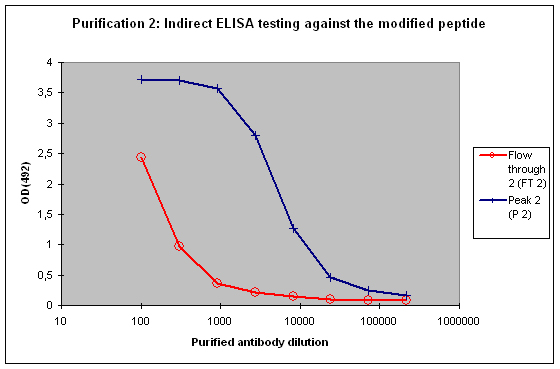
Figure 4 : Testing of Peak 2 (P2) and flow through 2 (FT2) against the modified peptide by indirect ELISA. Antibodies separated with the non-modified peptide react also with the modified peptide – these antibodies cannot be considered as a control for the non-modified protein target in your experimentation. The flow through 2 (FT2) antibodies show a weak reactivity against the modified peptide – please consider that the modification in the peptide might directly interact with the ELISA plate surface, and therefore a restricted access of antibodies to the modified epitope might result, causing a hyperbolical curve shape.
Please be advised that the population of antibodies against the modification in the deciding fraction FT2 can be very low, due to narrowing the variety of specific molecules towards a single feature of a small epitope. But nonetheless, the antibodies can be used in the most common application types diluted in the same way like other polyclonal antibodies.
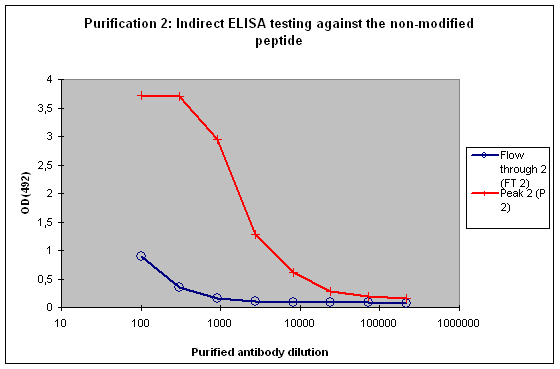
Figure 5 : Testing of Peak 2 (P2) and flow through 2 (FT2) against the non-modified peptide by indirect ELISA. P2 contains the majority of antibodies to the non-modified peptide, which is reflected by the strong reactivity of the antibodies to the target. FT2 might contain a small population also specific for the non-modified peptide. These antibodies can be diluted out or washed off in down stream applications, the soon drop of the curve from FT2 indicates that these antibodies are no good binders.
Please contact us if you wish to discuss your ELISA results in detail, or if you wish support for the establishment of your antibody.
Antibody Evolution (meaningful for 87-day immunization)
Eurogentec offers you the possibility to have ELISA testing done – this can be chosen as option for any immunization program. Simply ask for a complete program including ELISA right at the start (Please note, ELISA testing represents for 28-day Super Speedy immunization a set of supplementary characterisation data – the test can for technical reasons not be performed to decide on programme extension or stop).
In case ELISA testing with your program is ordered, we carry out the same standard program however without sending the peptide and sera directly to the customer. We keep the free peptide, the preimmune and small test sample (obtained after 3 injections) until the large bleed sample is available (day 66, after the 4th injection). At this moment, we carry out the ELISA test using one 96-well micro titre plate per animal. We test in parallel per ELISA dilutions from:
- Preimmune (PPI),
- Small (PP) and
- Large (GP) test bleed
Against
- The free peptide and
- Against the carrier protein,
- Including positive and negative controls.
The test report is sent to you per e-mail, and this is as well announced in the program related immunization schedule.
This paralleled testing not only allows to have a good view of the antibody evolution with the last boost, but it also gives a relatively good decision facilitation for program prolongation with additional boosts and bleeds, termination as scheduled, or immediate termination (Example 1).
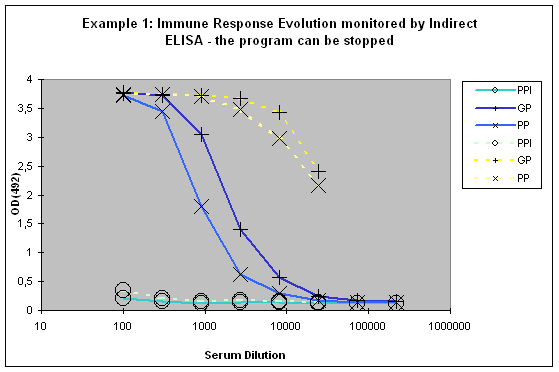
Figure 6 : In this anti-peptide program the immune responses against the peptide (solid lines) and against the carrier protein (dashed lines) are determined by Indirect ELISA (Coated antigen, one host, and three bleeds, referenced as AS-ELIS-01) and compare the reactivity of the serum before immunization (pre-immune serum, PPI, day 0), the small test bleed (PP, day 38), and the large bleed (GP, day 66). The success of the program is reflected by the sigmoidal curve shape of the plot, which evolved from “no response” (PPI), already strong responses after day 38 (PP), and the strongest responses after day 66 (GP). The program in this host can be finished, if desired, since the response evolution reached already at day 66 a maximum being characteristic for day 87 (the end point of a conventional 87-day program).
If a good increase is observed between the first and the second test bleed, the chances to get still higher titres with additional injections are quite good (Example 2). If however both bleeds (first and second test bleed) show more or less the same titre, an additional injection will most of the time result in only slight or no titre increase of specific antibodies.
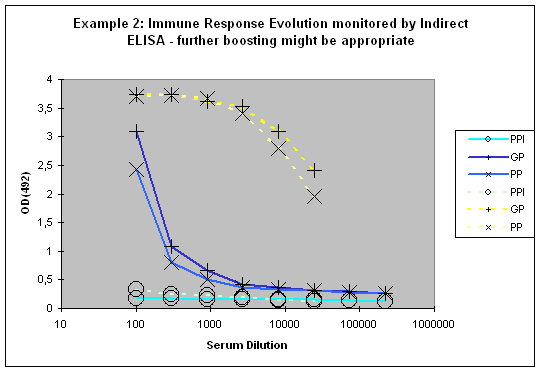
Figure 7 : In this anti-peptide program the immune responses against the peptide (solid lines) and against the carrier protein (dashed lines) are determined by Indirect ELISA (Coated antigen, one host, and three bleeds, referenced as AS-ELIS-01) and compare the reactivity of the serum before immunization (pre-immune serum, PPI, day 0), the small test bleed (PP, day 38), and the large bleed (GP, day 66). The antibody evolution of the program is slow, and reflected by the hyperbolic curve shape for the PP and GP for the peptide (blue) of the plots. The carrier (yellow, compare also to Fig. 1) produced already strong responses, which evolved from “no response” (PPI), to almost maximum at day 38 (PP), and the strongest responses after day 66 (GP). The program in this host should be extended by additional boosts in order to receive a sigmoidal curve shape (compare Fig.: 1, PP and GP, blue) reflecting the desired antibody reactivity against the antigen (Compare to blue curves PP and GP in Fig.: 1).
Please be advised that ELISA results are telling the customers and us if the immunization worked. Nonetheless, this information can also bear weaknesses. Usually proteins bind very good to ELISA plates but especially small antigens like peptides or other haptens might
- Not bind at all (a false negative result)
- Bind insufficiently (the result might partially reflect the immune response which has been evolved in the animal)
- Bind to the plate with by the epitope (a false negative result)
Please note the general efficiency in case of a peptide immunization program is reflected by the strong immune response to the carrier protein (a global positive control, Fig.: 1 and 2, yellow curves). If the response against the peptide appears to be strikingly weaker (Fig.: 2 for example; Fig.: 1 reflects rather the ideal case), you should in each case test the serum in your intended application, or by a different method, like dot or Western blotting.
Please note, it is hardly possible to extrapolate results from working ELISA tests to other applications, or to conclude from an antibody’s efficiency in one application to its potential capabilities in other applications: this means that an antibody providing bad results in ELISA testing, might be excellent in your down stream application, where the antigen looks different – but also vice versa is possible.
Please contact us if you further wish to discuss your ELISA results.
General considerations:
Please be advised that the antigen binding characteristics of an antibody after affinity purification might be shifted from polyclonal to a monoclonal-like specificity. A protein as antigen has much more epitopes for antibodies than a peptide. It is therefore in a few instances likely that especially in the case of peptides that key epitopes get hidden on ELISA plates, and ELISA results do not reflect properly the detection capabilities of the tested antibody, like they might be revealed in your intended application.
Eurogentec works with standardised protocols for purification of antibodies. Though these protocols are optimised, it might still be possible in rare cases that antibodies are not completely eluted from the purification column, nor can it be excluded that all partially denatured antibodies fold back to functional molecules during neutralisation after elution. In these cases affinity purification might not be the right way to concentrate your antibody.
Please note that immunization hosts are not transparent, and very rare occurring failure of immunization programs are not foreseeable to us, Eurogentec cannot guaranty for any antibody efficiency, nor warrant any quantities or capabilities of the produced molecules.
Which antigen formats are compatible with an immunization?
Lyophilized
You can send us your freeze-dried antigen at ambient temperature. If the antigen is poorly soluble in aqueous solution, we suggest that you avoid lyophilization and that you send us your protein in solution on dry ice. The addition of adjuvant will help to dissolve even lipophilic proteins. If the antigen does not dissolve, a fine suspension of the antigenwill be obtained by thorough mixing. Such fine suspensions can also be useful for antibody production because particles are efficiently phagocyted.
SDS-PAGE**
For antibody productions with SDS-PAGE gel fragments, we advise our customers to cut out the band of interest and to aliquot it in separate tubes for each injection. The antigen tubes can be shipped at room temperature. The standardCoomassie and Coomassie-like staining procedure can be used since the Coomassie staining dyes do not interfere with the antibody evolution. However, sylver stain is not allowed.
The band should just be washed briefly but thoroughly in water to remove acetic acid and methanol residues, and then cut into injection pieces, and aliquoted wet into safe lock tubes to avoid drying. The gel must not be dried or lyophilized, because this would make the fragmentation before injection more difficult.
** Not available for Speedy programme or small rodents.
In Solution
You can send us your antigen in solution on dry ice. We recommend limiting as far as possible the use of detergents and aggressive chemicals such as acetic acid, guanidine hydrochloride, heavy metals and other agents that are toxicto the host animal.
It is possible to immunize animals with an antigen solution containing 8M-urea, but this is more painful for the rabbits.For this reason, we ask our customers to send us the antigen as concentrated as possible so that we can dilute the solution before injection in order to decrease the final urea concentration. Antigens in solution should be sent in avolume not exceeding the authorized amount per injection.
e.g.:
- Rabbit 500 µL/injection
- Rat 250 µL/injection
- Guinea Pig 250 µL/injection
- Mouse 150 µL/injection
- Hen 500 µL/injection
- Goat/Sheep 100 µL/injection.
How should I store my serum?
Storage Suggestion for Antibodies and Serum used for in vitro Research Applications
Properly stored, antibodies should remain stable for months or years. Freezers with automatic de-frosting are absolutely inappropriate for antibody and serum storage.
If you are using catalogue antibodies, please refer to the provided data sheet.
Storage Temperatures
Our purified antibodies are delivered to you in PBS with 0,1% BSA as stabiliser and 0,01% Thimerosal as preservative. For these formulations we suppose that storage of small aliquots at –20°C is appropriate.
The aliquoting of antibodies has the following advantages:
- Minimisation of the damage due to freezing and thawing
- Exclusion of contamination due to multiple pipetting from a single vial
Aliquots should be thawed once, with any remaining antibody kept at 4°C.
When you receive an antibody you should carefully thaw it (in the fridge in a 4°C water bath – do not forget about it) and centrifuge it with 10.000 x g for 20 sec in order to pull down all the solution. Aliquot the antibody in low-protein-binding micro centrifuge tubes for storage. Please adapt the size of the aliquots to your typical experimental consumption. Aliquots should not be smaller than 10 µl to avoid damage of the antibody by evaporation and adhesion to the vessel wall.
Though it is considered in general as optional, we recommend the addition of 50% glycerol (v/v, final) to your antibodies for additional freezing and evaporation protection for storage at –20°C (glycerol is frozen at –80°C).
Please use just sterile chemicals and solutions for your antibodies to prevent down stream microbial growth.
Upon receipt, antibody storage for one or two weeks (time required for experimental establishment) at 4°C should be acceptable with subsequent aliquoted freezing for long-term storage.
Ascites and serum represent an exception here since the contained proteases require immediate storage at -20°C or -80°C.
Please be advised that sera produced at Eurogentec do not contain any preservative, 0,01% azide or thimerosal (depending on your research purpose) should be added to the aliquots for storage at –20°C after first thawing.
Egg yolks in contrast must never be frozen, since they denature into a form that does not allow isolation of IgY and affinity purification anymore. Egg Yolks from Eurogentec are shipped at 4°C with 0,01% azide added as preservative ready for short-term storage (over several weeks) at 4°C.
For long-term storage we recommend the purification of IgY from the egg yolks.
IgY isolates and affinity purified IgY antibodies should be treated like purified antibodies described in the beginning of this section.
Antibody formulations that require special attention:
- Enzyme-conjugated antibodies should not be frozen at all in order to retain the maximum enzymatic activity, storage at 4°C is therefore mandatory, please refer to the technical datasheet on the solvent formulation, and add if required 0,01% Thimerosal (Azide will inactivate HRP-conjugates)
- All conjugated antibodies (enzymes, fluorophores, or biotin) should be stored in the dark (very well suited: the black foil bags of x-ray films or photographic paper, and the corresponding boxing)
- Fluorophore-conjugated antibodies can be stored aliquoted at –20°C with 50% glycerol (final v/v) – please make sure by the data sheet that they are formulated as well with BSA, physiologically concentrated buffer salts (e.g. PBS), and preservatives (azide or thimerosal), furthermore, please be sure to use a sterile glycerol formulation, since microbial growth might occur and destroy the antibody. Storage at –80°C of glycerol containing solutions is not advised since this is below the freezing point of glycerol
- Minimisation of aggregate formation of some IgG isotypes (IgG3 for instance) requires storage at 4°C
Contamination prevention
Purified custom-made antibodies from Eurogentec already contain 0,01% thimerosal to avoid microbial growth, as mentioned on the data sheet. But please be advised that any serum, like Test Sera for animal selection, pre-immune sera (PPI), large bleed sera (GP), or final bleed sera (SAB) do not contain any preservative.
Please be advised of the following prior to use sodium azide for storage
- Azide is toxic to organisms and should be avoided in applications which are done with living cells, or tissues (the same is the case for thimerosal – please advise us before starting your immunization programme of your research purpose in order to avoid any preservative)
- If antibodies should be conjugated or spotted involving amino groups, azide will interfere negatively into the process, thimerosal is an acceptable alternative without these unwanted side effects
Preservatives, if already in an antibody formulation, can be removed in the following ways:
- By dialysis or centrifugal filtration: using a device with a MWCO of 14 kDa, please refer to the manufacturer’s recommendations for most efficient use centrifugal filter devices or dialysis equipment (MW of sodium azide ~ 65 D, thimerosal ~ 404 D, IgG ~ 160 kDa, IgM ~ 600 kDa).
- By gel filtration (which is faster than dialysis). Please refer to the manufacturer’s recommendations for most efficient use of suited filter cartridges
please be advised that all materials used here should be sterile to ensure the subsequent stability of the antibody, and to avoid contamination by purification
Damage caused by freezing and thawing of antibodies
The antibody’s binding capacity can be reduced by aggregate formation due to denaturation following cycles of freezing and thawing.
Though a lot of researchers keep their antibodies at –80°C, there is no real explanation why –20°C might be inappropriate. In any case, antibodies should rather be stored protected from daily routine in the freezer, e.g. rather in the back than in the front, or in drawers than standing free accessible.
Working concentrations of antibodies should not be stored for more than one day at 4°C, since proteins are more susceptible to degradation if they are stored in lower concentrations. Ideal would be to formulate the antibody with additional protein, like BSA or milk powder to reach concentrations of total protein in the mg/ml range. BSA or milk powder in working solutions also minimizes antibody loss due to attachment to the vessel wall.
The addition of stabilizing proteins is not recommended for antibodies that are subject to conjugation or array spotting.
What are the authorized buffer components for immunization?
Compound or formulations
| Buffer | Allowed | Not allowed |
| Water | Yes, keep the volume small (1 ml) | - |
| PBS | Yes, keep the volume small (1 ml) | - |
| Physiological buffer solutions |
Yes, keep the volume small (1 ml) | - |
| Metal dyes/heavy metals | - | Risk of toxicity |
| Salts (KCl, NaCl, MgCl2 | < 1.0 M | > 1.0 M |
| SDS | < 2.0 % | > 2.0 % |
| Urea | < 6.0 M | > 8.0 M |
| Guanidinium HCl | - |
Risk of toxicity |
| Digoxin/Digoxigenin | - | Risk of toxicity |
| Octylglucoside | <1.0 % | > 1.0 % |
| Triton X-100/Tween-20 | < 0.2 % | > 0.2 % |
| Glycerol | < 20 % | > 20 % |
| PMSF | - | Risk of toxicity |
| Pefabloc | < 0.1 mM | > 0.1 mM |
| Leupeptin/Pepstatin | < 1 μM | > 1 μM |
| DTT | < 3 M | > 3 M |
| Mercaptoethanol | - | Risk of toxicity |
| Imidazole | < 3 M | > 3 M |
| TFA | - | High risk of toxicity |
How to provide my antigen?
Lyophilized
You can send us your freeze-dried antigen at ambient temperature. If the antigen is poorly soluble in aqueous solution, we suggest that you avoid lyophilization and that you send us your protein in solution on dry ice. The addition of adjuvant will help to dissolve even lipophilic proteins. If the antigen does not dissolve, a fine suspension of the antigen will be obtained by thorough mixing. Such fine suspensions can also be useful for antibody production because particles are efficiently phagocyted.
SDS-PAGE**
For antibody productions with SDS-PAGE gel fragments, we advise our customers to cut out the band of interest and to aliquot it in separate tubes for each injection. The antigen tubes can be shipped at room temperature. The standard Coomassie and Coomassie-like staining procedure can be used since the Coomassie staining dyes do not interfere with the antibody evolution. However, Sylver stain is not allowed. The band should just be washed briefly but thoroughly in water to remove acetic acid and methanol residues, and then cut into injection pieces, and aliquoted wet into safe lock tubes to avoid drying. The gel must not be dried or lyophilized, because this would make the fragmentation before injection more difficult.
** Not available for Speedy program or small rodents.
In solution
You can send us your antigen in solution on dry ice. We recommend limiting as far as possible the use of detergents and aggressive chemicals such as acetic acid, guanidine hydrochloride, heavy metals and other agents that are toxic to the host animal. It is possible to immunize animals with an antigen solution containing 8M-urea, but this is more painful for the rabbits. For this reason, we ask our customers to send us the antigen as concentrated as possible so that we can dilute the solution before injection in order to decrease the final urea concentration. Antigens in solution should be sent in a volume not exceeding the authorized amount per injection.
For ex:
- Rabbit 500 µL/injection
- Rat 250 µL/injection
- Guinea Pig 250 µL/injection
- Mouse 150 µL/injection
- Hen 500 µL/injection
- Goat/Sheep 100 µL/injection