Antibodies additional services
Antigen design & selection
A high antigen quality is key to generate efficient antibodies. A careful selection, design and production of the most convenient antigen will allow generating the best antibody for your application.
Choosing the best antigen
The best antigen will mimic as closely as possible the molecule which is the target of the antibody in the final application.
The target is a protein
If the target is a protein, the best antigen is certainly the protein itself. Any difference between the antigen and the natural target will result in an antibody perfectly matching the antigen, but not recognizing the target in its natural context.
Some important factor have to be taken into account:
What is the complexity of my antigen?
The antigen complexity is important, as it is directly proportional to the complexity of the antibody which will be generated. The most complex antigens will contain numerous epitopes, all being a potential target for the host’s immune system, which will generate as many different antibodies.
How can the host influence immunization?
Because each animal is different, they will all produce a different antibody mix, some epitopes being recognized as more dominant by the immune system of each host, hence more targeted. Collectively, the polyclonals obtained from individual immunizations should recognize the complex antigen very efficiently.
What is the impact on my application?
But if the final assay targets one specific epitope only, it is likely that the polyclonal obtained from one immunization will perform better compared to another one. In this case, using a more specific antigen better matching the antibody application may be useful.
Advantages of using a peptide as antigen
Specific antigens like peptides contain a few different epitopes.
Specificity
Accordingly, a polyclonal generated from a peptide immunization will be more specific than a polyclonal obtained using the whole protein as the antigen.
Protein detection
An anti-peptide antibody may perfectly detect the corresponding protein, provided that there is not sequential, conformational, or post-translation modification difference between the peptide and the protein region from which it originates.
Helping you in selecting the best antigen
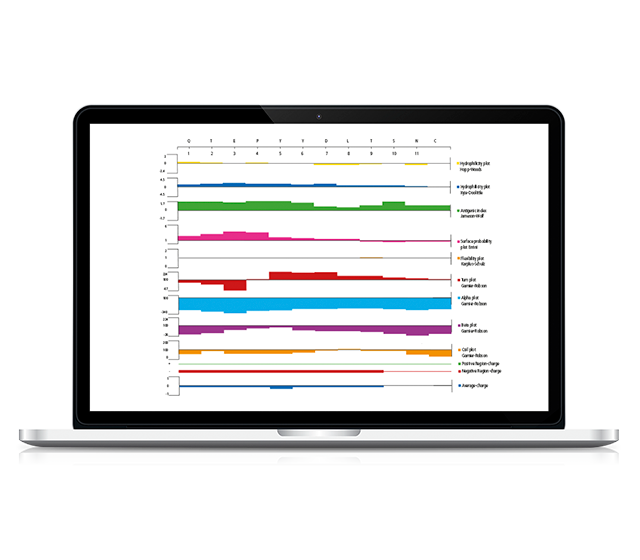
Customized bioinformatics studies may be used to help selecting the best antigen, considering critical features:
- Antigen drift
- Homology with non-relevant proteins
- B epitopes prediction
- Protein structure analysis
- …
Our team of experts is available to helping you.
Using a protein as the antigen
Overview
When a protein is to be used as an antigen, it is most often produced recombinantly. Care must be taken that the recombinant and the native protein forms can differ. For example, the protein in its native environment may be associated to other protein partners, which may hide dominant epitopes not accessible to the antibody in a cellular context. Also, the protein may be post-translationally modified, while the recombinant protein may not be.
The antigen can be:
Inclusion bodies
When producing proteins in E. coli, inclusion bodies are obtained in many cases. Within inclusion bodies, the protein retains some secondary structures, but the tertiary structure is not folded. Using inclusion bodies as the antigen is practically possible, but the antibody fraction which targets non-natural epitopes within the inclusion bodies will for sure not recognize the protein in its 3D from.
Depending on the protein, and the application, the antibody fraction which targets epitopes common to the inclusion bodies and the folded protein may be good enough to allow detecting the protein in its natural environment.
Urea-denatured protein
Obtaining a correctly folded protein from inclusion bodies requires a denaturation/renaturation process which is strictly protein-dependent, and always challenging. Urea is often used as the denaturing agent. But incubating a protein with urea for prolonged periods will lead to carbamylation at the N-termini of proteins/peptides and at the side chain amino groups of Lys and Arg residues.
Therefore, using a protein with urea as the antigen will generate antibodies against these non-natural epitopes. Because Lys and Arg residues are often exposed on the protein surface, part of the polyclonal antibodies generated with this strategy will fail binding to the native protein form. This limitation must be carefully considered before using urea solutions to generate antibodies.
Cuanidium hydrochloride (GuHCl)-denatured protein
GuHCl is a powerful protein denaturing agent, which does not lead to carbamylation as urea. But GuHCl is toxic and cannot be injected in animals. If a protein denatured with GuHCl is to be used as an antigen, dialysis will be a pre-requisite.
Denatured protein
If one wants to use a denatured protein as the antigen, injecting an SDS-PAGE gel fragment may be an option. The limit here is that this cannot apply to small rodents and is not compatible with a Speedy 28-days program.
We produce your recombinant protein
- Full-length proteins, Protein fragments, Poly-epitopes, Fusion proteins, And more…
- Bacterial and eukaryotic host cells
- Denaturation/Renaturation of proteins produced as inclusion bodies
- Purification
- Optimization of production yields on request