Additional services
Antibody Purification
Depending on the antibody type and its application, the format may vary from crude serum up to highly purified antibody. At Eurogentec, we handle several purification methods using the best strategies and equipment’s.
IgG is the most common antibody isotype produced in mammals, while IgY is found in majority in birds. Protein A and protein G can bind with more or less affinity and specificity to the Fc region of IgG antibodies from mammals. Purifying IgY’s requires specific pretreatments.
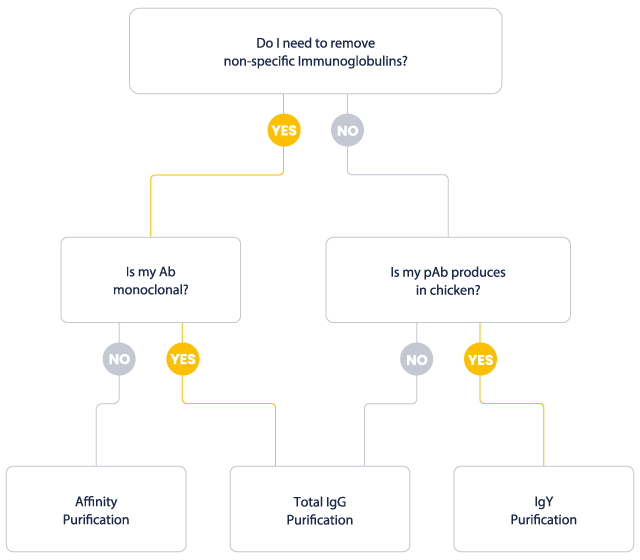
Which purification for which application?
Polyclonal antibodies are often delivered as crude serum, corresponding the host’s blood from which red blood cells and clotting factors have been removed. The serum of an immunized host contains immunoglobulins from all classes, 10% of which are considered as antigen-specific. When the non-antigen specific antibodies interfere with the assay, the crude serum is purified. In some applications, using the crude serum gives satisfactory results, and a purification is not mandatory.
At Eurogentec, monoclonal antibodies are exclusively produced in vitro. Culture media with high, low or no serum are used, and a total immunoglobulin purification usually follows.
Affinity purification aims at separating the antigen-specific antibodies from the non-specific fraction. The affinity chromatography technique is employed for this purpose, in which the unique biological specificity of the analyte and ligand interaction is utilized for the separation.
The antigen is immobilized on the stationary phase*, and the serum or medium containing the antibody is applied onto the column.
The affinity-purified antibody is suitable for many applications.
*It is important to note that the antigens compatible with an affinity purification must be free from many additives, such as high salt concentrations, denaturing agents (urea, guanidium hydrochloride), glycerol, preservatives (sodium azide, etc), as they may interfere with the binding process of the antigen to the stationary phase. Therefore, although some additives are tolerated for the immunization phase, they may be excluded when the same antigen is to be used for affinity purification
In the particular case of hen immunization to generate specific IgY antibodies, a particular process must take place, because the IgY migrate to the egg yolk, which is highly charged with lipids and lipoproteins. A specific extraction process must be applied prior to the affinity purification.
Total IgG purification relies on the high affinity of Protein A (from Staphylococcus aureus) or Protein G (from Streptococcus) for the Fc domain of Immunoglobulins. When applied to polyclonal antibodies, protein A/G purification removes the serum proteins, but does not eliminate the non-specific immunoglobulin fraction. Accordingly, a protein A/G purified serum may still generate some cross-reactivities in the final assay. When applied to monoclonal antibodies, the purification efficiency is higher, because the serum-containing medium is less charged in contaminating immunoglobulins compared to crude serum.
IgY are produced in hen egg ylod. They cannot be purified with Protein A or G. IgY are isolated with a particular purification process.
Discover our purification services
Affinity purification
The amount of antigen which is necessary depends on the serum volume to purify.
|
Antigen Amount |
Serum Volume to purify |
|---|---|
| 3 mg antigen (peptide or protein) | 5 mL to 10 mL |
| 6 mg antigen (peptide or protein) | 20 mL |
| 8 mg antigen (peptide or protein) | 50 mL |
The purified antibodies are delivered in a PBS buffer containing 0.01% thimerosal and 0.1% BSA. Should another buffer be used, please let us know.
Starting from 20 mL serum purification, the ELISA test and High-Resolution electrophoresis analysis are included in the service, for more information contact us.
IgY purification
The isolation of IgY from the egg yolk and their subsequent affinity purification are labor-intensive. Eurogentec had developed high quality extraction and purification IgY processes as an option to the chicken immunization programs. A minimum of 2 egg yolks are processed.
Total IgG purification
Whole IgG purification may be performed on Protein A or Protein G, depending on the host species. For example, rat antibodies do not bind Protein A, while guinea pig antibodies are better purified using Protein A.
Binding capacities of immunoglobulins from different species to protein A and G
| Origin of Immunoglobulins | Protein A | Protein G |
|---|---|---|
| Mouse | +++ | ++++ |
| Rat | - | +++ |
| Guinea pig | ++++ | ++ |
| Rabbit | ++++ | +++ |
| Goat | +/- | ++ |
| Sheep | - | ++ |
| Pig | +++ | +++ |
| Horse | ++ | ++++ |
| Hen (IgY) | - | - |
High-Resolution electrophoresis analysis is provided with the purified serum.