Antibody additional services
Antibody fragmentation
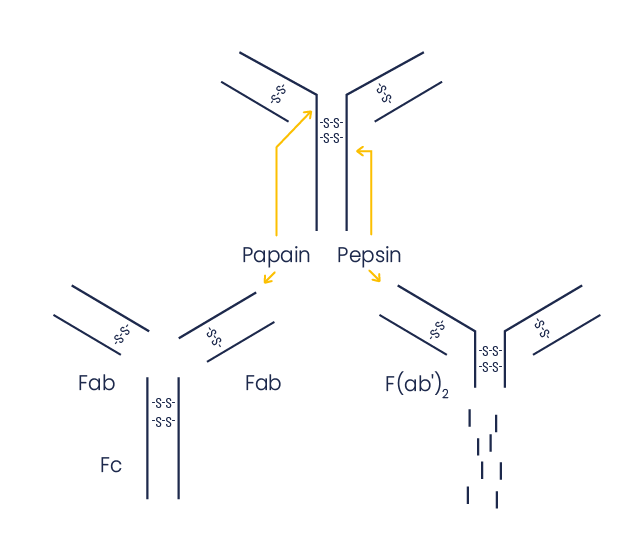
Using full-size antibodies, which are large and complex proteins, may be limiting for some assays. When the application requires, having access to antibody fragments may be an advantage. Eurogentec selectively cleaves IgG’s to generate Fab or F(ab’)2 fragments.
Generating Fab and F(ab’)2 antibody fragments
Both Fab and F(ab’)2 are antigen-binding fragments which contain the variable regions of the heavy and light chains (VH and VL, respectively), associated by disulfide bonds to preserve the native conformation of the antigen-binding site.
The Fc domain does not bind the antigen; it contains some carbohydrates, which may be useful for antibody conjugation, and mediates cellular effector functions, as many cells have receptors for the Fc fragment leading to unspecific background.
Antibody fragmentation requires significant Ab amounts (about 10 mg) and is usually performed on monoclonals.
Fragmentation methods
Papain cleaves IgG’s above the hinge region containing the disulfide bridges which join the heavy chains, but below the site of disulfide bond formation between the heavy and light chains. This leads to an intact Fc fragment and two monovalent Fab fragments
Pepsin cleaves IgG’s below the hinge region containing the disulfide bridges which join the heavy chains, generating a bivalent F(ab′)2 fragment with two antibody binding sites. The Fc fragment is digested into small peptides
Advantages of small size of antibody fragments
Due to their small size, Fab and F (ab′)2 antibody fragments are suitable for in vivo applications as well as assays where the epitope accessibility is low. Due to their higher penetration into tissue sections, immunohistochemistry signals are improved.
Other advantages of antibody fragments
Because they lack the Fc domain, they do not bind to Fc receptors on the surface of multiple cells, including macrophages, monocytes, B-cells and NK cells. Using F (ab′) 2 or Fab fragments. This ensures that they will bind to their antigen only, with less background staining
The bivalent character of F(ab′)2 antibody fragments allows cross-linking experiments with two antigens and immunoprecipitations.